Intermediate Bulk Containers (IBCs) are a flexible, cost-effective materials handling solution for pharmaceutical manufacturing facilities.
But can they be used effectively in a building that only has a single floor available? Yes, they can!
Benefits of IBCs
The manufacture of pharmaceutical oral solid dosage (OSD) products is a tightly regulated, highly competitive industry, and it can be difficult to find ways to make cost savings without compromising on product quality or safety.
As a materials-handling solution for your powders or granules, Intermediate Bulk Containers (IBCs) are flexible, affordable, and eradicate many of the traditional sources of waste in a continuously coupled or manual production facility.

With IBCs, fewer ingredients are lost as escaped dust or residues and cross-contamination or problems with open air industrial mixing are all but eliminated.
Furthermore, different sized IBCs can be easily integrated into multiple, parallel production lines to reduce bottlenecks, and manufacture different products and batch sizes without reducing capacity.
But does an IBC-based pharmaceutical facility work if you only have a small or single-floor building? Wouldn’t a continuous, single-level production line make the most efficient use of space?
Using IBCs with a single-floor facility
Single floor facilities are among the cheapest to build or lease, but their size can limit what you are able to do with them. Often, smaller OSD manufacturers lean towards the traditional connected production line set-up, in which ingredients are transferred between process using a conveying system, moving along a continuously coupled system to emerge at the other end as finished products.
A problem with this type of set-up is that it is very inflexible. Having one process directly coupled to the next means it is very difficult to change recipes, batch sizes, or to operate parallel production lines in an efficient way, without investing in whole new set of equipment for multiple production suites.
The flexible nature of IBCs means that you can actually fit more into a smaller space, and restricted ceiling height or a single-level building doesn’t have to be a barrier.
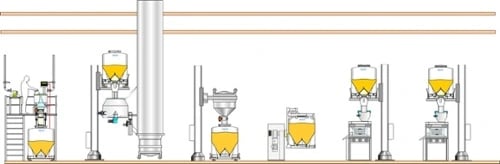
In single-floor configurations, IBCs can be used to connect one process to another by matching the volume of the IBC system to the batch volume – in other words, the IBC used should be of a sufficient size to achieve a full batch transfer. This has the advantage of requiring only one automated connection/disconnection per manufacturing step, and means that parallel processes can be operated, at their optimal speed and capacity, at the same time, on the same level.
Get creative - use gravity to your advantage
Though you may not have an ‘upstairs’ to your pharmaceutical facility, if your ceiling height allows, it is possible to make use of efficient vertical materials transfer methods such as gravity-fed systems. The solution here is to use pillar lifts to elevate the IBCs.

Safe, convenient, and far more cost effective than moving your whole manufacturing operation to new, bigger premises, installing a pillar lift or a framework around processing equipment can help you make the most of a small space.
Posted by
Matt BaumberTopics:
Pharmaceutical

